Preparation of titanium nitride(TiN)
Preparation of titanium nitride(TiN)
The chemical properties of nitrogen are very stable, but under some special conditions, it can form nitrides with many elements. Among these nitrides, the transition metal nitride-titanium nitride (TiN) has become the focus of research at home and abroad. Titanium nitride is a structural material with attractive golden yellow, high melting point, high hardness, good chemical stability, small wettability with metal, and high electrical conductivity and superconductivity. It can be applied to high-temperature structural materials and Superconducting materials.
1. The structure and properties of titanium nitride
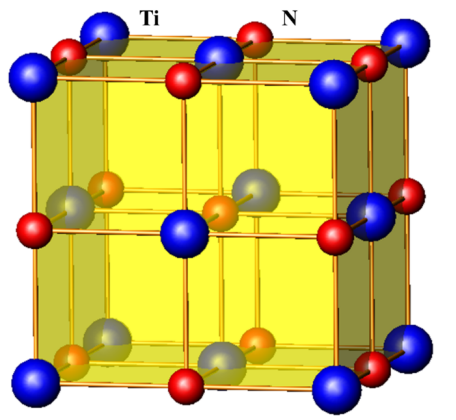
TiN has a typical NaCl-type structure and belongs to a face-centered cubic lattice. The top of the face-centered cubic is nitrogen atoms, and the titanium atoms are located in the (1/2,0,0) space position of the face-centered cubic. TiN is a non-stoichiometric compound, and its stable composition range is TiN0.6~TiN1.16, and the nitrogen content can be changed within a certain range without causing changes in the TiN structure. TiN powder is generally yellowish brown, ultrafine TiN powder is black, and TiN crystals are golden yellow. The lattice constant of TiN is a=4.23 nm, the lattice constant of TiC is a=4.238 nm, and the lattice constant of TiO is a=4.15 nm. The lattice parameters of these three substances are very close, so the nitrogen in the TiN molecule Atoms can be replaced by oxygen and carbon atoms at any ratio to form a solid solution. The physical and chemical properties of titanium nitride are determined by the content of nitrogen. When the content of nitrogen is reduced, the lattice parameters of titanium nitride will increase, and the hardness will also increase. The microscopic increase, but the seismic resistance of titanium nitride decreases.
The physical properties of titanium nitride:
melting point 2950.6~3205.8℃, linear expansion coefficient of 5.712~7.053×106(1/K)(25℃), density of 5.435~5.447g/cm3, thermal conductivity of 25.081(W·m -1·K-1) (300~2000℃), the Mohs hardness is 8-9. In general, the color of titanium nitride powder is yellow-brown, the color of black is ultrafine titanium nitride powder, the color of yellow is titanium nitride crystal, and a large number of aggregated titanium nitride crystals have golden yellow metallic luster.
The chemical properties of titanium nitride:
It is relatively stable and does not react with water and acids (hydrochloric acid and sulfuric acid), but it can be incorporated into hydrofluoric acid. If the hydrofluoric acid contains an oxidant, the titanium nitride will be completely dissolved in hydrogen Fluoric acid. When titanium nitride is dissolved in a strong alkaline solution, it decomposes and releases ammonia gas.
2.Preparation of Titanium Nitride
2.1 Direct nitriding of titanium powder or TiH2
Nitriding titanium powder in nitrogen or hydrogen atmosphere at 1273~1673K for 1~4h, repeat the operation several times after the product is crushed, and obtain stoichiometric titanium nitride powder. The equation is:
2Ti+N2=2TiN
The metal hydride TiH2 can also be used for nitriding, which can be reacted below 1273K. The equation is:
2TiH2+N2=2TiN+2H2
The advantage of this method is that it is easy to operate and can obtain high-quality titanium nitride powder, but the disadvantage is that the raw material price is too high to be mass-produced, and this process is prone to powder sintering, resulting in losses.
2.2 TiO2 carbothermal reduction nitridation method
The carbothermic reduction and nitridation method of TiO2 uses TiO2 as a raw material and carbon graphite as a reducing agent to react with N2 to generate TiN. The synthesis temperature is 1380~1800℃, and the reaction time is about 15h. In this reaction environment, carbon not only reacts with oxygen, but also reacts with titanium to form TiC, because the lattices of titanium carbide, titanium nitride and titanium oxide are very close, and the three are easy to form a solid solution.
The TiN obtained by this method is generally not high in purity, and the content of O and C is relatively high. In order to obtain TiN with a relatively low content of O and C, a higher reaction temperature and a longer reaction time are required. In addition, some experts also use another method, that is, the magnesium powder and titanium oxide are mixed and reacted in a certain proportion under the condition of lower temperature to prepare titanium nitride.
2.3 Microwave carbothermic reduction method
The microwave carbothermic reduction method is an oxidation-reduction reaction that uses inorganic carbon as a reducing agent at a higher temperature. Domestic Liu Binghai et al. used this method to prepare titanium nitride powder. The specific operation is as follows: using titanium oxide as a raw material, heating carbon with microwaves until the temperature reaches 1200°C, and maintaining the reduction reaction at this temperature for 1 hour to obtain titanium nitride powder. Compared with the conventional method, the titanium nitride powder prepared by this method has higher purity, and has the advantages of low synthesis temperature (100-200°C lower than the original) and short cycle (1/15 of the conventional method).
2.4 Chemical vapor deposition method
The chemical vapor deposition method uses gaseous TiCl4 as a raw material, H2 as a reducing agent and N2 to generate TiN, and the synthesis temperature is 1100~1500℃. Coatings on the surface of metals and ceramics often use this process to enhance the hardness and wear resistance of ceramics and metals. The synthesized TiN has high purity, but low production efficiency and high cost. This process is a common method for coating TiN film on the surface of metal, ceramics and other items to make it beautiful
2.5 Self-propagating high-temperature synthesis method
The self-propagating high-temperature synthesis method is also called the combustion synthesis method. This method is to directly ignite the titanium powder (in the form of a blank) in nitrogen (restrict a certain pressure), and then burn the titanium powder in nitrogen to obtain TiN products. This process has been extensively researched and commercialized in Russia, the United States, and Japan.
According to domestic research reports in this area, Wang Weimin et al. used this process to prepare TiN ceramic powder, and studied the influence of process parameters such as green compact density, diluent, and nitrogen partial pressure on the synthesis. Liu Suying and others are also investigating this process.
2.6 Mechanical alloying method
The mechanical alloying method is to place titanium powder in an ammonia or nitrogen system, and use a high-energy ball mill to make them interact under the strong collision and agitation of the grinding balls to obtain nano-titanium nitride. This is a brand-new synthesis method. In China, Liu Zhijian and others used TiH1.924 powder instead of Ti powder to react with nitrogen. Using this high-energy ball milling process, after high-energy ball milling in flowing ammonia for 100h, almost all TiH1.924 is converted to TiN, and the conversion rate is obtained. A great improvement. And Zhou Li and others later used the same method to prepare nano-titanium nitride powder, and the reaction time was only 9h.
2.7 Molten salt synthesis method
The molten salt synthesis method has not been reported in the preparation of titanium nitride, but it is a good research direction to study the preparation of titanium nitride by this method. This method uses a low melting point molten salt as the reaction medium. The reactants can be dissolved in the molten salt. The entire reaction is completed in an atomic-level environment. After the reaction is completed, the salts can be dissolved and filtered with a suitable solvent. Get the product. The product obtained by this method has high purity, simple operation, short reaction time, no harsh requirements on the reaction temperature, easy control of the morphology and particle size of the product, and no agglomeration.
2.8 Sol-gel method
The sol-gel method is to mix the reactants uniformly under liquid phase conditions, and then undergo the process of hydrolysis and condensation. The reactants form a transparent sol in the solution. The sol forms a gel after aging and slow polymerization. After the glue is dried and cured, the material we need is obtained.
The chemical properties of nitrogen are very stable, but under some special conditions, it can form nitrides with many elements. Among these nitrides, the transition metal nitride-titanium nitride (TiN) has become the focus of research at home and abroad. Titanium nitride is a structural material with attractive golden yellow, high melting point, high hardness, good chemical stability, small wettability with metal, and high electrical conductivity and superconductivity. It can be applied to high-temperature structural materials and Superconducting materials.
1. The structure and properties of titanium nitride
TiN has a typical NaCl-type structure and belongs to a face-centered cubic lattice. The top of the face-centered cubic is nitrogen atoms, and the titanium atoms are located in the (1/2,0,0) space position of the face-centered cubic. TiN is a non-stoichiometric compound, and its stable composition range is TiN0.6~TiN1.16, and the nitrogen content can be changed within a certain range without causing changes in the TiN structure. TiN powder is generally yellowish brown, ultrafine TiN powder is black, and TiN crystals are golden yellow. The lattice constant of TiN is a=4.23 nm, the lattice constant of TiC is a=4.238 nm, and the lattice constant of TiO is a=4.15 nm. The lattice parameters of these three substances are very close, so the nitrogen in the TiN molecule Atoms can be replaced by oxygen and carbon atoms at any ratio to form a solid solution. The physical and chemical properties of titanium nitride are determined by the content of nitrogen. When the content of nitrogen is reduced, the lattice parameters of titanium nitride will increase, and the hardness will also increase. The microscopic increase, but the seismic resistance of titanium nitride decreases.

The physical properties of titanium nitride:
melting point 2950.6~3205.8℃, linear expansion coefficient of 5.712~7.053×106(1/K)(25℃), density of 5.435~5.447g/cm3, thermal conductivity of 25.081(W·m -1·K-1) (300~2000℃), the Mohs hardness is 8-9. In general, the color of titanium nitride powder is yellow-brown, the color of black is ultrafine titanium nitride powder, the color of yellow is titanium nitride crystal, and a large number of aggregated titanium nitride crystals have golden yellow metallic luster.
The chemical properties of titanium nitride:
It is relatively stable and does not react with water and acids (hydrochloric acid and sulfuric acid), but it can be incorporated into hydrofluoric acid. If the hydrofluoric acid contains an oxidant, the titanium nitride will be completely dissolved in hydrogen Fluoric acid. When titanium nitride is dissolved in a strong alkaline solution, it decomposes and releases ammonia gas.
2.Preparation of Titanium Nitride
2.1 Direct nitriding of titanium powder or TiH2
Nitriding titanium powder in nitrogen or hydrogen atmosphere at 1273~1673K for 1~4h, repeat the operation several times after the product is crushed, and obtain stoichiometric titanium nitride powder. The equation is:
2Ti+N2=2TiN
The metal hydride TiH2 can also be used for nitriding, which can be reacted below 1273K. The equation is:
2TiH2+N2=2TiN+2H2
The advantage of this method is that it is easy to operate and can obtain high-quality titanium nitride powder, but the disadvantage is that the raw material price is too high to be mass-produced, and this process is prone to powder sintering, resulting in losses.
2.2 TiO2 carbothermal reduction nitridation method
The carbothermic reduction and nitridation method of TiO2 uses TiO2 as a raw material and carbon graphite as a reducing agent to react with N2 to generate TiN. The synthesis temperature is 1380~1800℃, and the reaction time is about 15h. In this reaction environment, carbon not only reacts with oxygen, but also reacts with titanium to form TiC, because the lattices of titanium carbide, titanium nitride and titanium oxide are very close, and the three are easy to form a solid solution.
The TiN obtained by this method is generally not high in purity, and the content of O and C is relatively high. In order to obtain TiN with a relatively low content of O and C, a higher reaction temperature and a longer reaction time are required. In addition, some experts also use another method, that is, the magnesium powder and titanium oxide are mixed and reacted in a certain proportion under the condition of lower temperature to prepare titanium nitride.
2.3 Microwave carbothermic reduction method
The microwave carbothermic reduction method is an oxidation-reduction reaction that uses inorganic carbon as a reducing agent at a higher temperature. Domestic Liu Binghai et al. used this method to prepare titanium nitride powder. The specific operation is as follows: using titanium oxide as a raw material, heating carbon with microwaves until the temperature reaches 1200°C, and maintaining the reduction reaction at this temperature for 1 hour to obtain titanium nitride powder. Compared with the conventional method, the titanium nitride powder prepared by this method has higher purity, and has the advantages of low synthesis temperature (100-200°C lower than the original) and short cycle (1/15 of the conventional method).
2.4 Chemical vapor deposition method
The chemical vapor deposition method uses gaseous TiCl4 as a raw material, H2 as a reducing agent and N2 to generate TiN, and the synthesis temperature is 1100~1500℃. Coatings on the surface of metals and ceramics often use this process to enhance the hardness and wear resistance of ceramics and metals. The synthesized TiN has high purity, but low production efficiency and high cost. This process is a common method for coating TiN film on the surface of metal, ceramics and other items to make it beautiful
2.5 Self-propagating high-temperature synthesis method
The self-propagating high-temperature synthesis method is also called the combustion synthesis method. This method is to directly ignite the titanium powder (in the form of a blank) in nitrogen (restrict a certain pressure), and then burn the titanium powder in nitrogen to obtain TiN products. This process has been extensively researched and commercialized in Russia, the United States, and Japan.
According to domestic research reports in this area, Wang Weimin et al. used this process to prepare TiN ceramic powder, and studied the influence of process parameters such as green compact density, diluent, and nitrogen partial pressure on the synthesis. Liu Suying and others are also investigating this process.
2.6 Mechanical alloying method
The mechanical alloying method is to place titanium powder in an ammonia or nitrogen system, and use a high-energy ball mill to make them interact under the strong collision and agitation of the grinding balls to obtain nano-titanium nitride. This is a brand-new synthesis method. In China, Liu Zhijian and others used TiH1.924 powder instead of Ti powder to react with nitrogen. Using this high-energy ball milling process, after high-energy ball milling in flowing ammonia for 100h, almost all TiH1.924 is converted to TiN, and the conversion rate is obtained. A great improvement. And Zhou Li and others later used the same method to prepare nano-titanium nitride powder, and the reaction time was only 9h.
2.7 Molten salt synthesis method
The molten salt synthesis method has not been reported in the preparation of titanium nitride, but it is a good research direction to study the preparation of titanium nitride by this method. This method uses a low melting point molten salt as the reaction medium. The reactants can be dissolved in the molten salt. The entire reaction is completed in an atomic-level environment. After the reaction is completed, the salts can be dissolved and filtered with a suitable solvent. Get the product. The product obtained by this method has high purity, simple operation, short reaction time, no harsh requirements on the reaction temperature, easy control of the morphology and particle size of the product, and no agglomeration.
2.8 Sol-gel method
The sol-gel method is to mix the reactants uniformly under liquid phase conditions, and then undergo the process of hydrolysis and condensation. The reactants form a transparent sol in the solution. The sol forms a gel after aging and slow polymerization. After the glue is dried and cured, the material we need is obtained.











(1).png)

.jpg)

